- Messages
- 13
- Location
- Stockton on tees, England
Hello
I always use RO with Treated Tap Water only because i was told a long time ago that you should have some sort of mineral content in your tanks.
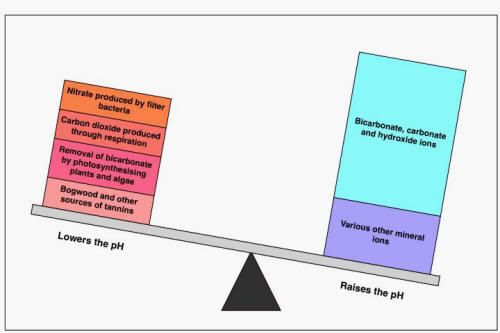
I know that if i want to bring the KH down to lower the PH you use Ro or by some other means if need be.
But recently i have come accross some people who only use pure RO.
What i want to know from you all out there is do you do the same or do you belive that a bit of mineral content in yer tank goes a long way to the health of your fish.
Please discuss.
Regards
Phil Wood
I always use RO with Treated Tap Water only because i was told a long time ago that you should have some sort of mineral content in your tanks.
I know that if i want to bring the KH down to lower the PH you use Ro or by some other means if need be.
But recently i have come accross some people who only use pure RO.
What i want to know from you all out there is do you do the same or do you belive that a bit of mineral content in yer tank goes a long way to the health of your fish.
Please discuss.
Regards
Phil Wood